+COVERAGE+MECHANISM+OF+ACTION%3A.jpg)
Moya B, Zamorano L, Juan C, Ge Y, Oliver A (2010) Affinity of the new cephalosporin CXA-101 to penicillin-binding proteins of Pseudomonas aeruginosa. Page MGP, Bush K (2014) Discovery and development of new antibacterial agents targeting Gram-negative bacteria in the era of pandrug resistance: is the future promising? Curr Opin Pharmacol 18:91–97 Clin Microbiol Rev 26(4):792–821ĭrawz SM, Papp-Wallace KM, Bonomo RA (2014) New beta-lactamase inhibitors: a therapeutic renaissance in an MDR world. Pucci MJ, Bush K (2013) Investigational antimicrobial agents of 2013. Nat Rev Microbiol 12:231–232īutler MS, Blaskovich MA, Cooper MA (2013) Antibiotics in the clinical pipeline in 2013. Rex JH (2014) ND4BB: addressing the antimicrobial resistance crisis. Theuretzbacher U (2012) Accelerating resistance, inadequate antibacterial drug pipelines and international responses. Kostyanev T, Bonten MJ, O’Brien S, Steel H, Ross S, Francois B, Tacconelli E, Winterhalter M, Stavenger RA, Karlén A, Harbarth S, Hackett J, Jafri H, Vuong C, MacGowan A, Witschi A, Angyalosi G, Elborn JS, deWinter R, Goossens H (2015) The Innovative Medicines Initiative’s New Drugs for Bad Bugs programme: European public-private partnerships for the development of new strategies to tackle antibiotic resistance. World Health Organization, Genevaīrown ED (2013) Is the GAIN Act a turning point in new antibiotic discovery? Can J Microbiol 59(3):153–156 WHO (2015) Global action plan on antibiotic resistance. Clin Infect Dis 48(1):1–12ĬDC (2013) Antibiotic resistance threats in the United States, 2013.
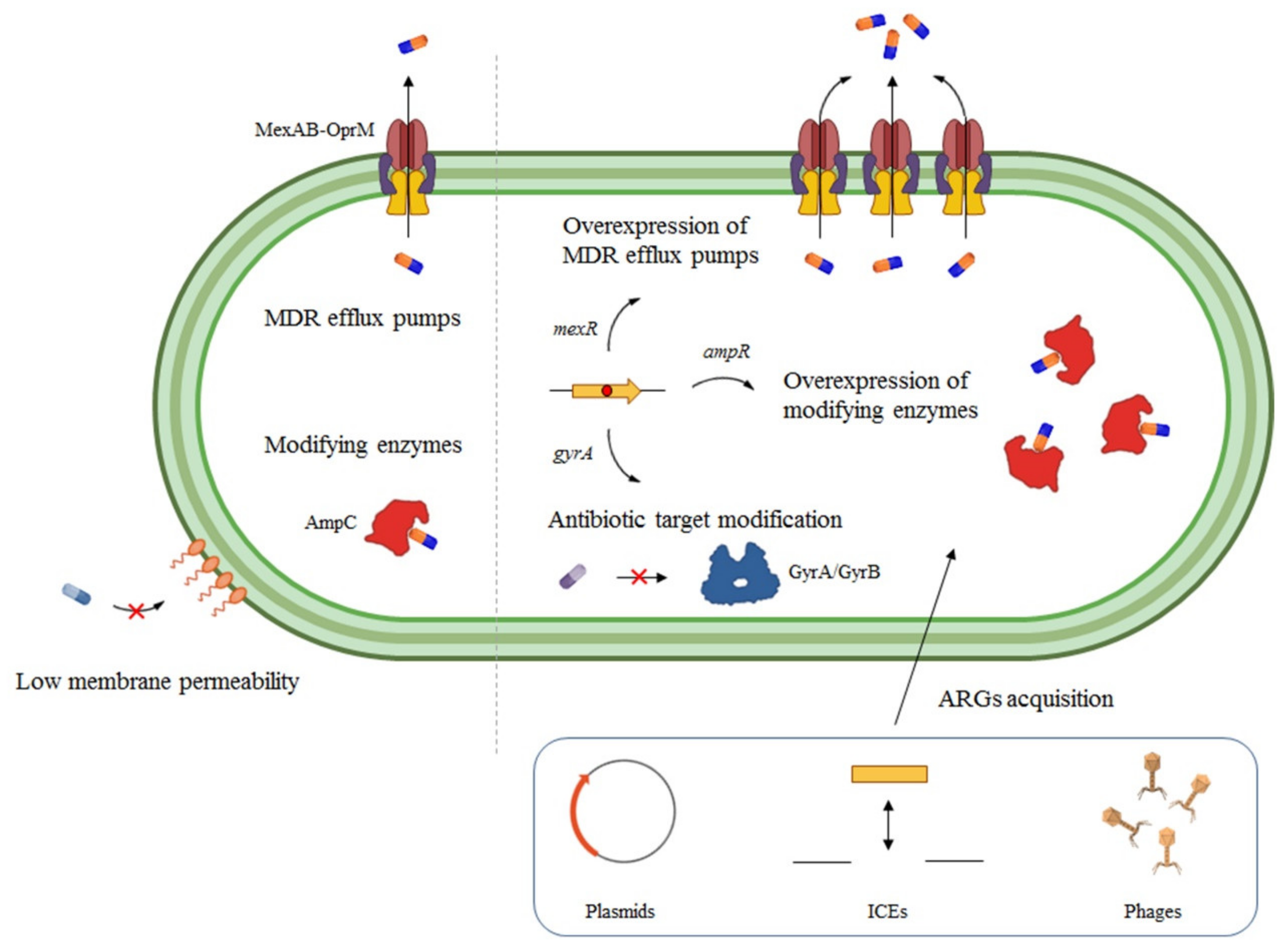
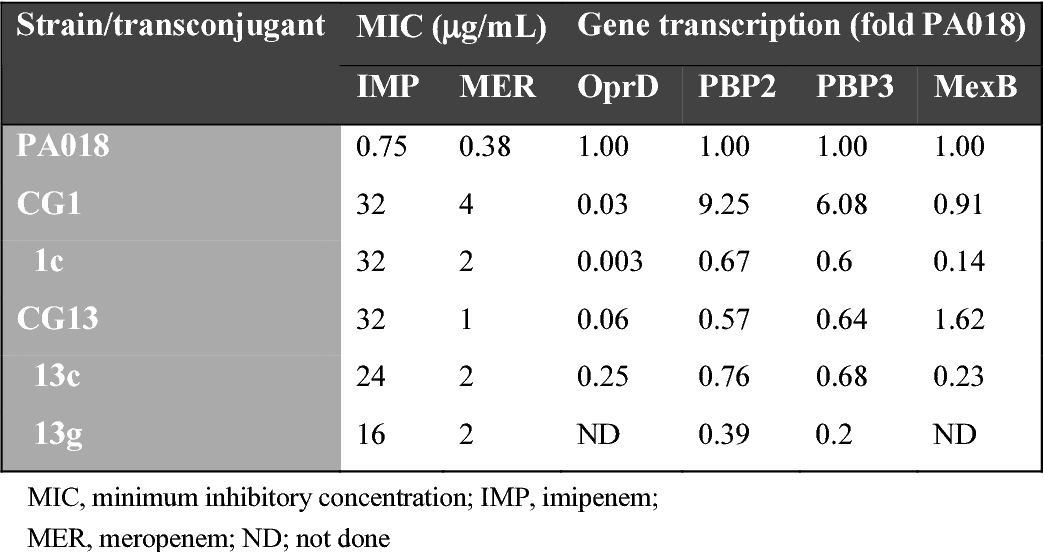
#Aztreonam ki pseudomonas aeruginosa pbp3 update
Clin Infect Dis 56(12):1685–1694īoucher HW, Talbot GH, Bradley JS, Edwards JE, Gilbert D, Rice LB, Scheld M, Spellberg B, Bartlett J (2009) Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin Pharmacol Ther 96(2):166–168īoucher HW, Talbot GH, Benjamin DK, Bradley J, Guidos RJ, Jones RN, Murray BE, Bonomo RA, Gilbert D (2013) 10 × ’20 Progress-development of new drugs active against Gram-negative bacilli: an update from the Infectious Diseases Society of America. Tomayko JF, Rex JH, Tenero DM, Goldberger M, Eisenstein BI (2014) The challenge of antimicrobial resistance: new regulatory tools to support product development.
Projan SJ, Shlaes DM (2004) Antibacterial drug discovery: is it all downhill from here? Clin Microbiol Infect 4:18–22 The potential impact of resistance mechanisms on the new agents is assessed and the ways in which PK/PD studies are used to design dosing regimens for the new agents, especially combinations, as well as to improve dosing of existing antibiotics are discussed. Although the new analogues of existing classes, and novel combinations, have been designed to address specific resistance problems, it is by no means certain than they will not be affected by the general mechanisms of resistance, particularly decreased net flux across the Gram-negative outer membrane. There are 6 molecules representing novel classes of antibiotics but only one of these, murepavadin, is expected to have activity against a Gram-negative pathogenic bacterium ( Pseudomonas aeruginosa). The combination of such inhibitors, which have broad activity against serine β-lactamases and may even inhibit some penicillin binding proteins, with carbapenems, cephalosporins or aztreonam, provides enhanced activity against multi-drug resistant Gram-negative bacteria. In addition, there is considerable activity around the discovery of novel classes of β-lactamase inhibitors with 10 combinations representing 4 inhibitor classes, at different stages of development. The majority of these are representatives of established antibiotic classes that have been modified to address problems of resistance. There are some 43 small molecules in the antibiotic development pipeline from late preclinical stage (7 compounds) through Phase 1 (11 molecules), Phase 2 (13 molecules) to Phase 3 (12 molecules).
